Natural gas condensate
Natural gas condensate is a low-boiling mixture of hydrocarbon liquids that are present as gaseous components in the raw natural gas produced from many natural gas fields.[1]
If the raw gas temperature is reduced to below its hydrocarbon dew point, the liquids begin to condense out of the gas. The resulting condensate is also referred to as simply condensate, or gas condensate, or sometimes natural gasoline because it contains hydrocarbons within the gasoline boiling range.
Raw natural gas may come from any one of three types of gas wells:[2][3]
- Crude oil wells – Raw natural gas that comes from crude oil wells is called associated gas. This gas can exist separate from the crude oil in the underground formation, or dissolved in the crude oil.
- Dry gas wells – These wells typically produce only raw natural gas that does not contain any hydrocarbon liquids. Such gas is called non-associated gas.
- Condensate wells – These wells produce raw natural gas along with low-boiling hydrocarbon liquids. Such gas is also non-associated gas and often referred to as wet gas.
Composition of natural gas condensate
There are hundreds of wet gas fields worldwide and each has its own unique gas condensate composition. However, in general, gas condensate has a specific gravity ranging from 0.5 to 0.8 and may contain:[1][4][5][6]
- Hydrogen sulphide (H2S}
- Thiols traditionally also called mercaptans (denoted as RSH, where R is an organic group such as methyl, ethyl, etc.)
- Carbon dioxide (CO2)
- Straight-chain alkanes having from 2 to 12 carbon atoms (denoted as C2 to C12)
- Cyclohexane and perhaps other naphthenes
- Aromatics (benzene, toluene, xylenes and ethylbenzene)
Separating the condensate from the raw natural gas
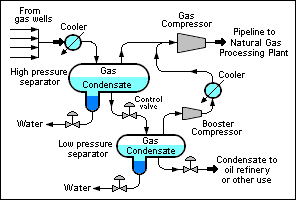
There are many different equipment configurations for the processing required to separate natural gas condensate from a raw natural gas. The schematic flow diagram to the right depicts just one of the possible configurations.[7]
The raw natural gas feedstock from a gas well or a group of wells is cooled to lower the gas temperature to below its hydrocarbon dew point. That condenses a good part of the gas condensate hydrocarbons. The resulting mixture of gas, liquid condensate and water is then routed to a high pressure separator vessel where the water and the raw natural gas are separated and removed. The raw natural gas from the high pressure separator is sent to the main gas compressor.
The liquid condensate from the high pressure separator flows through a throttling control valve to a low pressure separator. The reduction in pressure across the control valve causes the condensate to undergo a partial vaporization referred to as a flash vaporization. The raw natural gas from the low pressure separator is sent to a "booster" compressor which raises the gas pressure and sends it through a cooler and on to the main gas compressor. The main gas compressor raises the pressure of the gases from the high and low pressure separators to whatever pressure is required for the pipeline transportation of the gas to the raw natural gas processing plant. The main gas compressor discharge pressure will depend upon the distance to the raw natural gas processing plant and it may require that a multi-stage compressor be used.
At the raw natural gas processing plant, the gas will be dehydrated and acid gases and other impurities will be removed from the gas. Then the ethane (C2), propane (C3), butanes (C4) and C5 plus higher molecular weight hydrocarbons (referred to as C5+) will also be removed and recovered as byproducts.
The water removed from both the high and low pressure separators will probably need to be processed to remove hydrogen sulfide before the water can be disposed of or reused in some fashion.
Some of the raw natural gas may be re-injected into the gas wells to help maintain the gas reservoir pressures.
Characterizing the amount of condensate in natural gas
In the United States of America, the hydrocarbon dew point of processed, pipelined natural gas is related to and characterized by the term GPM, meaning gallons of liquifiable hydrocarbons per million (106) cubic feet of natural gas at a stated temperature and pressure. When the liquifiable hydrocarbons are characterized as being hexane or higher molecular weight components, they are reported as GPM (C6+) meaning hydrocarbons with 6 carbon atoms or more.[8][9]
However, it should be noted that the quality of raw produced natural gas (before it is purified by processing) is also often characterized by the term GPM, meaning the gallons of liquifiable hydrocarbons contained in a thousand (103) cubic feet of the raw natural gas. In such cases, when the liquifiable hydrocarbons in the raw natural gas are characterized as being ethane or higher molecular weight components, they are reported asGPM (C2+). Similarly, when characterized as being propane or higher molecular weight components, they are reported asGPM (C3+).[8][9]
Care must be taken not to confuse the two different definitions of the term GPM.
References
- ↑ Jump up to: 1.0 1.1 Francis S. Manning and Richard E. Thompson (1991). Oilfield Processing of Petroleum (Volume One:Natural Gas). Pennwell Books. ISBN 0-87814-343-2.
- ↑ International Energy Glossary (a page from the website of the Energy Information Administration)
- ↑ Natural gas processing (a page from the website of the Energy Information Administration)
- ↑ Natural Gas Condensate Kinder-Morgan MSDS
- ↑ Natural Gas Condensate Marathon Oil Company MSDS
- ↑ Natural Gas Condensate (Sour) Amerada Hess Corporation MSDS
- ↑ Simplified Process Flow Diagram
- ↑ Jump up to: 8.0 8.1 White Paper on Liquid Hydrocarbon Drop Out in Natural Gas Infrastructure (NGC+ Liquid Hydrocarbon Dropout Task Group, October 15, 2004)
- ↑ Jump up to: 9.0 9.1 White Paper on Liquid Hydrocarbon Drop Out in Natural Gas Infrastructure (NGC+ Liquid Hydrocarbon Dropout Task Group, September 28, 2005)