Aspergillus flavus: Difference between revisions
imported>Iqra Oza No edit summary |
imported>Iqra Oza No edit summary |
||
| Line 51: | Line 51: | ||
Aspergillus flavus by Constituents of Walnut (Juglans regia). Journal of Agricultural and Food Chemistry. 2004, 52, 1882-1889</ref> | Aspergillus flavus by Constituents of Walnut (Juglans regia). Journal of Agricultural and Food Chemistry. 2004, 52, 1882-1889</ref> | ||
'''Effect of Nigella sativa Extract and Oil on Aflatoxin Production by Aspergillus Flavus | '''Effect of Nigella sativa Extract and Oil on Aflatoxin Production by Aspergillus Flavus''' | ||
The effect of the crude exract of Nigella sativa, N sativa oil, ground coffee beans and caffeine were tested on A. flavus afltatoxin production. Nigella sativa and N. sativa oil are tested because N. sativa has many medicinal and antimicrobbial effects. Ground coffee beans and caffeine were tested because caffeine has been shown to inhibit both aflatoxin production and fungus growth. In this experiment local A. flavus strains were first isolated. Then the effects of each was tested using peanuts. They tested the effects of different amounts of crude N. sativa extract, N. sativa oil, caffeine and locally roasted ground coffee. They tested by adding ten grams of powdered peanuts to a one hundred mililiter BHI (brain-heart infusion) broth with the added variable( the extract, oil, coffee beans or caffeine). The broth was then sterilized and then inoculated with the fungus. Crude N. sativa extract inhibited the growth of aflatoxin G1, B1, and B2 at five percent concentration. At 1.25% concentration it only inhibited G1 and and at 2.5% concentration G1 and B2 were inhibited. N. sativa oil concentrated at three percent inhibited all aflatoxin production. But at one and two percent it did not inhibit B1 and B2. At two percent and four percent coffee bean concentration inhibit G1 and G2 aflatoxin production. At six percent coffee bean concentration inhibits B1 G1 and G2 aflatoxin production. Caffein inhibits only G1 and G2 at two, four and six percent concentration. > sativa oil was the most effective followed by N. sativa extract in aflatoxin inhibition. The things tested can be used in processed food to prevent foods contaminated with A. flavus from producing aflatoxins. <ref name=oil> Maraqa, Anwar. Al-Sharo'a, Najwa F., Farah, Husni. Elbjeirami, Wafa M. Shakya, Ashok K.,Sallal, Abdul-Karim J. Effect of Nigella sativa Extract and Oil on Aflatoxin Production by Aspergillus flavus. Turkey Journal of Biology, 31, (2007) 155-159 </ref> | |||
Revision as of 21:30, 13 May 2009
For the course duration, the article is closed to outside editing. Of course you can always leave comments on the discussion page. The anticipated date of course completion is May 21, 2009. One month after that date at the latest, this notice shall be removed. Besides, many other Citizendium articles welcome your collaboration! |
| Aspergillus Flavus | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
 | ||||||||||||||
| Scientific classification | ||||||||||||||
| ||||||||||||||
| Binomial name | ||||||||||||||
| Aspergillus flavus |
Description and significance
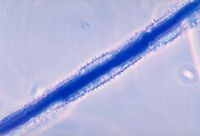
Aspergillus flavus is a haploid filamentous fungi. It produces a mycotoxin called aflatoxin B that is carcinogenic.[1] It causes liver damage and is one of the mycotoxins used as a biologcal weapon[2]. When crops are infected with A. flavus their value decreases because of the potential human or animal exposure to the aflatoxin.[3] A. flavus is mainly a saprophyte, meaning it obtains its nutrients from dead or decaying material, but it can also be a pathogen to plants and animals including humans.[1]
Pathology
A. flavus is an opportunistic pathogen, which allows it to infect animals, humans and plants. It is not virulent in healthy viable tissues. A. flavus targets seeds with poor viability. It infects seeds by entering through wounds and holes from which insect larval have exited, and by entering from the vascular tissue. A. flavus can infect plants through thier seeds or by being carried to the surface of the plant by insects or wind. Also it can be colonized but not infected until the plant is matured. A. flavus infects cotton by entering from natural openings and traveling up into the boll. In the boll the fungus can be there for twenty five days, not infecting the boll until matures. In peanuts the fungus can infect seeds as well as the nuts by penetrating the pods that peanuts grow in.[4]
In animals including humans A. flavus is associated with aflatoxicosis. Symptoms of aflatoxicosis in humans are abdominal pain, vomiting, pulmonary edema, convulsions, coma, liver damage, and death. In animals symptoms include liver damage, death, anemia, immune suppression, gastrointestinal dysfunction, a decrease in milk production, egg production, reproduction, and feed consumption. In Humans who eating foods with low amounts of aflatoxin over a long period of time can cause cancer. Eating foods with high amounts of aflatoxin can cause acute or chronic toxicity. Symptoms of chronic toxicity include decrease in growth rate, lowered milk or egg production, immunosuppression, jaundice and a swollen gall bladder. [5] When A. flavus is inhaled it can cause aspergillosis, a secondary respiratory infection in immuno-compromised patients. Aspergillosis can cause illness in three ways, pulmonary aspergillosis is an allergic reaction in people who have asthma, as a fungus ball called aspergilloma in scarred lung tissue or as infections that spread to other parts of the body by the blood stream.[5] Infections A. flavus can cause are corneal, otomycotic, and nasoorbital infections. [6]
Ecology
Aspergillus flavus can be found all over the world; however it is found to be abundant in places of warm temperature. Field contamination is mostly present in areas with high temperatures and drought. It is also abundant in areas with temperate climates during warm drought years. In the US it affects the corn crop in the south eastern area.[4]
A. flavus grows in temperatures of 25-42˚C, but the optimal temperature for growth is 37˚C. It survives through winter as a mycelium or sclerotium. Sclerotium is a resistant structure that can develop into hyphae or conidia. The conidia are then scattered into air and soil by insects or wind.[1]
Cell structure and metabolism
A. flavus belong to the phylum Ascomycota. Ascomycota can reproduce sexually by forming ascospores, but A. flavus does not produce ascospores. Since it does not produce ascopores is can only reproduce asexually.[7] A. flavus is also known as a mold. Like other molds it also grows by producing hyphae. The network of hyphae, or mycelia,are responsible for secreting catabolic enzymes. The enzymes secreted are used to break down complex food sources. The complex food sources are broken down into small molecules which are then absorbed by the mycelium, which use the small molecules to grow. Mycelia produce asexual spores called conidia. Both the mycelia and conidia can be seen with the naked eye while the individual hyphae cannot. Young conidia in A. flavus are yellow green and as the spores age they turn a darker green. A. flavus can use many nutrient sources but being a saphrophyte it uses primarily dead or decaying materials.[1] Being a saphrophyte is important for A. flavus life cycle because it allows the fungus to be present in the soil with decaying plant tissue, allowing the A. flavus to infect other plants. [5]
Genome structure
The genome was sequenced at The Institute for Genomic Research in Maryland. It has also been entered into the BLAST database.[1]The genome of A. flavus strain NRRL 3357 has been sequenced. It has approximately 13,487 genes and the average length of each gene is 1485 bp.[1]
Application to Biotechnology
With the sequencing of the genome done we know that it is
Current Research
With the sequencing of the genome done current research is focused on using the sequence to understand the fungus, to yield crops that are resistant to the fungus and to determine tools to control the production of the aflatoxin. There have been may unsuccessful experiments conducted on yielding A. flavus resistant crops of peanuts and corn. Currently research is being done to cultivate genes that are resistant to A. flavus afflatoxin production. These genes will then be used to create resistant crops.
Phytochemical Inhibition of Aflatoxigenicity in Aspergillus flavus by Constituents of walnut
Tulare walnuts are more resistant to aflatoxin formation especially when compared to Chico walnuts. Researchers were trying to find the phytochemical constituents from Tulare walnuts that are capable of inhibiting aflatoxigenesis or colonization of A. flavus. Researchers collected both Tulare and Chico walnuts every two weeks. They started collecting in June early in the season, and stopped in early September when the nuts were mature. Nuts were collected from Wolfskill Experimental Orchard in California. The orchard had two trees of each Tulare and Chinco walnuts. For ten nuts of each variety the pellicle was separated from the kernel by hand. Nut kernels and pellicle were then ground up and passed through a 1 mm scree with a Resch model ZM-1 mill. Gallic acid, ellagic acid, and tannic acid were found. These three were then added to a medium, the control in this experiment was a medium without any of the acids. The fungus was added to each medium and after incubation the fungus was analyzed for aflatoxin. The walnut pellicles were analyzed for their ability to inhibit aflatoxin production. Researchers found that factors of resistance resided completely in the pellicle. In the pellicle the walnut has gallic acid, which can inhibit activity toward aflatoxin production. Regulation of galic acid in walnuts by either conventional breeding or genetic manipulation can lead to new crops with high resistance to aflatoxigenisis. [8]
Effect of Nigella sativa Extract and Oil on Aflatoxin Production by Aspergillus Flavus
The effect of the crude exract of Nigella sativa, N sativa oil, ground coffee beans and caffeine were tested on A. flavus afltatoxin production. Nigella sativa and N. sativa oil are tested because N. sativa has many medicinal and antimicrobbial effects. Ground coffee beans and caffeine were tested because caffeine has been shown to inhibit both aflatoxin production and fungus growth. In this experiment local A. flavus strains were first isolated. Then the effects of each was tested using peanuts. They tested the effects of different amounts of crude N. sativa extract, N. sativa oil, caffeine and locally roasted ground coffee. They tested by adding ten grams of powdered peanuts to a one hundred mililiter BHI (brain-heart infusion) broth with the added variable( the extract, oil, coffee beans or caffeine). The broth was then sterilized and then inoculated with the fungus. Crude N. sativa extract inhibited the growth of aflatoxin G1, B1, and B2 at five percent concentration. At 1.25% concentration it only inhibited G1 and and at 2.5% concentration G1 and B2 were inhibited. N. sativa oil concentrated at three percent inhibited all aflatoxin production. But at one and two percent it did not inhibit B1 and B2. At two percent and four percent coffee bean concentration inhibit G1 and G2 aflatoxin production. At six percent coffee bean concentration inhibits B1 G1 and G2 aflatoxin production. Caffein inhibits only G1 and G2 at two, four and six percent concentration. > sativa oil was the most effective followed by N. sativa extract in aflatoxin inhibition. The things tested can be used in processed food to prevent foods contaminated with A. flavus from producing aflatoxins. [9]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 Aspergillus flavus hosted by aspergillusflavus.org
- ↑ [Mold-Help.org. "Aspergillus."] hosted by Moled-help.org
- ↑ [Mellon J, Cotty P, Dowd M. Aspergillus flavus hydrolases: their roles in pathogenesis and substrate utilization. Applied Microbiology & Biotechnology [serial online]. December 2007;77(3):497-504. Available from: Academic Search Premier, Ipswich, MA. Accessed April 12, 2009]
- ↑ 4.0 4.1 [KLICH M. Aspergillus flavus: the major producer of aflatoxin. Molecular Plant Pathology [serial online]. November 2007;8(6):713-722. Available from: Academic Search Premier, Ipswich, MA. Accessed April 15, 2009]
- ↑ 5.0 5.1 5.2 [1] hosted by pathport.vbi.vt.edu
- ↑ Accessed April 13, 2009 hosted by wikipedia.com
- ↑ [2]
- ↑ Mahoney, Noreen and Molyneux, Russel J.Phytochemical Inhibiion of Aflatoxigenicity in Aspergillus flavus by Constituents of Walnut (Juglans regia). Journal of Agricultural and Food Chemistry. 2004, 52, 1882-1889
- ↑ Maraqa, Anwar. Al-Sharo'a, Najwa F., Farah, Husni. Elbjeirami, Wafa M. Shakya, Ashok K.,Sallal, Abdul-Karim J. Effect of Nigella sativa Extract and Oil on Aflatoxin Production by Aspergillus flavus. Turkey Journal of Biology, 31, (2007) 155-159