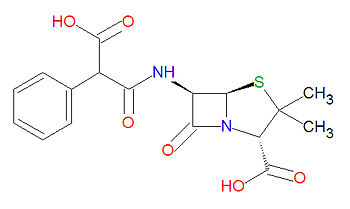
Carbenicillin
Carbenicillin, or carboxybenzylpenicillin, is a broad-spectrum beta-lactam-based antibiotic and is a derivative of penicillin. It is used for infections of the urinary tract. The most useful aspect of this drug is its antipseudomonal and antiproteal activity, but is used to treat a wide variety of bacterial infections in the urinary tract from bacteria including E. coli, Enterobacter species, Enterococci, Morganella morganii, Proteus mirabilis, Proteus vulgaris, Pseudomonas and [[{Providencia rettgeri]].
Mechanism of action
Like other beta-lactam penicillin derivatives, carbenicillin interference with final stage of bacterial cell wall synthesis by acylating the penicillin-sensitive transpeptidase enzyme's C-terminal domain by opening the lactam ring. This prevents cross-linking of peptidoglycan strands, and leads to autolysis of the bacterial cells by the autolysin enzymes.
Chemistry
Carbenicillin is a beta-lactam-based antibiotic, which acylates a transpetidase enyzme through a lactam ring-opening reaction. Its IUPAC chemical name is (2S,5R,6R)-6-[(3-hydroxy-3-oxo-2-phenylpropanoyl)amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid and it has chemical formula C17H18N2O6S (MW = 378.3996 g/mol).
External links
- Carbenicillin - FDA approved drug information (drug label) from DailyMed (U.S. National Library of Medicine).
- Drug Bank [1]