DNA
DNA (Deoxyribonucleic acid), a very large biological molecule (a chemical compound) found in the nucleus of almost every cell, is responsible for providing the genetic information necessary for the development and reproduction of all living organisms. Every living organism has its own unique DNA code which is stored in the DNA molecule as a sequence of smaller repeating molecules (a biopolymer) - much like a genetic 'barcode' or 'fingerprint'. DNA functions as a template, enabling the transfer of information from the sequenced genetic code within the DNA molecule to an enzyme (a biochemical assembly machine) called a ribosome, that translates the code into a protein molecule. The ribosome assembles a protein molecule from amino acids (the building blocks of proteins) according to the code's sequence. While every cell containing DNA in any one organism has identical DNA, each different cell type will synthesize the proteins common to most cells, along with an unique organization of proteins that defines the specialized functions of that particular cell type.
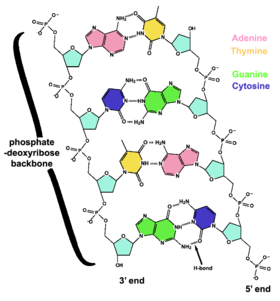
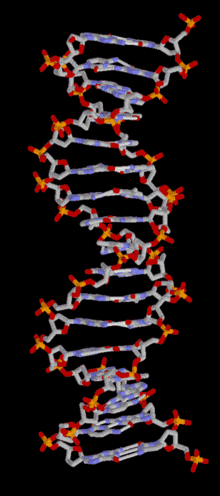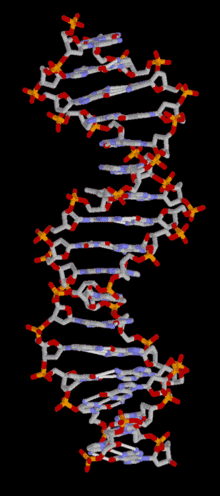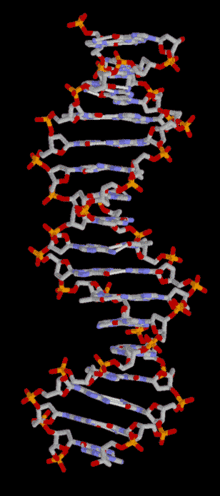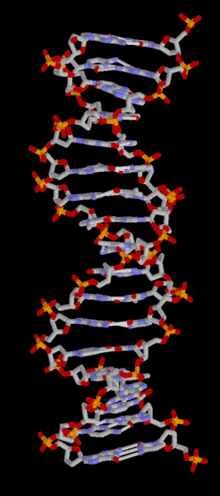
In most organisms, DNA is structured as double-helix formation consisting of two DNA strands coiled around each other in a head-to-toe "antiparallel" orientation. The strands provide a structural support for a complementary pair of bases. A base is like a letter of a word. A series of three base pairs forms a codon( a DNA word) on the DNA strand that encodes the information for one amino acid residue. A series of codons, and associated start/stop codons, (A DNA sentence) form the genetic code (gene) for the selection of particular amino acids and their specific arrangement necessary for the assembly of a protein molecule. The protein molecules, as many as 20,000 different types, are used in the cell, or are transported to other areas of the organism, important for intercellular communication.
Each single strand of DNA is a long biopolymer comprised of repeating units called nucleotides (a nucleotide is a base linked to a sugar and one or more phosphate groups)[1] which form a sugar/phosphate backbone. Attached to each sugar molecule (deoxyribose) is one of four bases; Adenine (A), Thymine (T), Guanine (G) or Cytosine (C). Each base is a structural complement of its opposing base; adenine always pairs with thymine and guanine always pairs with cytosine. The complementary base pairs are identical in size and shape and will fit between the backbones of double stranded DNA in only one of these four configurations - TA, AT, GC and CG. The complementary strands are held together by hydrogen bonds between the bases. This complementarity, at work in all the DNA functions, makes it possible for DNA to be copied and repaired relatively easily, while accurately preserving its information content and thereby forms the basis of semi-conservative DNA replication. Template:TOC-left Nuclear DNA is organized into chromosomes each containing many genes. At conception, the male sperm and female ovum (an egg) each contribute 23 chromosomes for a total of 46 chromosomes in the fertilized embryo. In eukaryotes (organisms such as plants, yeasts and animals whose cells have a nucleus) DNA is mostly stored inside the cell nucleus. Some organelles in eukaryotic cells (mitochondria and chloroplasts) have their own DNA with a similar organization to bacterial DNA. In prokaryote cells (organisms such as common bacteria), DNA is located in a region called the nucleoid. Viruses have a single type of nucleic acid, either DNA or RNA, directly encased in a protein coat called the capsid. Some cells such as blood cells do not contain DNA.
The entire DNA sequence of genes in any organism is called its genome, which provides the necessary genetic instructions to produce the phenotype, the outward physical manifestation of an organism.
Seeking the secret of life
Important genetic discoveries in DNA research spanning many decades included the surprising realization that the seemingly simple DNA molecule, and not the more complex protein molecule, is responsible for the internally coded, inheritible information defining the genotype. It was taken for granted that only protein was comples enough to contain genetic information. A further significant discovery was that the paired bases exist in equal proportions. This led to the discovery of the duplex structure of DNA, usually referred to as the "double-helix", that enables the complementary replication of DNA in living organisms. The most important feature of the duplex model is the introduction of the concept of complementarity. Complementarity has come to explain the entire sequence of events in the expression of genetic functions.[2]
Isolating genetic material
DNA was first isolated by Friedrich Miescher who, in 1869, discovered a microscopic substance in the pus of discarded surgical bandages. As it resided in the nuclei of cells, he called it "nuclein".[3] This discovery was followed by Phoebus Levene's identification of the base, sugar and phosphate nucleotide unit in 1929 .[4] Levene suggested that DNA consisted of a string of nucleotide units linked together through the phosphate groups. However Levene thought the chain was short and the bases repeated in a fixed order.
In 1928, Frederick Griffith demonstrates a "transforming factor" that can transmit the ability of Pneumococcus bacteria to cause pneumonia in mice. Although DNA had been identified in cells it was not considered a strong candidate for the genetic material due to its simple form. However, in 1943, Oswald Theodore Avery extended Griffith's work and discovered that traits of the virulent form of Pneumococcus could be transferred to the avirulent form of the same bacteria by mixing killed virulent bacteria with the live avirulent form. Avery identified DNA as the transforming factor.[5] This discovery is considered by some to be the major breakthrough enabling a new focus on DNA itself and represent the birth of molecular biology.[6]
DNA's role in heredity was confirmed in 1953, when Alfred Hershey and Martha Chase in the Hershey-Chase experiment, showed that DNA is is the genetic material of the T2 phage.[7] This experiment provided the crucial evidence that it was DNA itself, (which was shown to enter the cell), and not a protein,(which remained outside the cell), that was the carrier of information.
Discovering the double helix and code
In 1950, Erwin Chargaff published a paper in which he described the equal ratios of adenine to thymine, as well as cytosine to guanine, something he found extraordinary; he called this the complementary situation, but later it became known as the Chargaff Ratios. This discovery would play a crucial role in determining the structure of DNA. [8]
As early as 1937 William Astbury produced the first X-ray diffraction patterns that showed that DNA had a regular structure.[9] But it was not until 1953, based on the high quality X-ray diffraction images[10] taken by Rosalind Franklin and the information provided by Erwin Chargaff that the bases were paired, that James D. Watson and Francis Crick constructed a cardboard model that led to [10] what is now accepted as the first accurate model of DNA structure. Their discovery on the morning of February 28, 1953 was reported in the journal Nature.[11]
Experimental evidence for Watson and Crick's model were published in five articles in the same issue of Nature.[12] Of these, Franklin and Raymond Gosling's paper[13] saw the publication of the X-ray diffraction image [14], which was key in Watson and Crick interpretation, as well as another article, co-authored by Maurice Wilkins and his colleagues.[15] Franklin and Gosling's subsequent paper identified the distinctions between the A and B structures of the double helix in DNA.[16] In 1962 Watson, Crick, and Maurice Wilkins jointly received the Nobel Prize in Physiology or Medicine (Franklin didn't share the prize with them as she had died earlier).[17]
In an influential presentation in 1957, Crick laid out the "central dogma" of molecular biology, which foretold the relationship between DNA, RNA, and proteins, and articulated the "adaptor hypothesis".[18] Final confirmation of the replication mechanism that was implied by the double-helical structure followed in 1958 through the Meselson-Stahl experiment.[19] Further work by Crick and coworkers showed that the genetic code was based on non-overlapping triplets of bases, called codons, allowing Har Gobind Khorana, Robert W. Holley and Marshall Warren Nirenberg to decipher the genetic code.[20]
Big science: the Human Genome Project
Initially biologists could only clone and sequence genes one by one, which required large amounts of labor and time. As technology improved in the 1980's, some scientists such as Craig Venter and Leroy Hood believed that the focus could shift to sequencing whole genomes and they advocated a era of big science epitomised by the Human Genome Project (HGP). Their goal was to automate the mapping and sequencing to produce resources that gives science a valuable shortcut to addressing the biology of the genome.
Leroy Hood designed DNA sequencing machines to automate the process and thus massively reduce the cost of large scale sequencing. The chemistry of dideoxy sequencing devised by Frederick Sanger in 1977 was still the primary tool but new fluorescent labeling techniques, polymerase chain reaction (PCR) and genomic BAC/YAC libraries were technological advances that made automation possible. Craig Venter advocated a random "shotgun" approach to sequencing rather than the traditional and slower target driven approach. At first, this was in the form of sequencing a massive number of randomly picked expressed sequence tags (EST's). Each tag represented a cDNA clone and the whole collection represented a catalog of expressed genes in a genome. This approach transitioned to sequencing random BAC's from the whole genome and was coined shotgun sequencing.
The HGP started out as an academic pursuit that used a methodological tactic of establishing unique genetic markers, breaking the DNA into smaller bits which were then sequenced and, using the markers as a guide, reassembled. The public consortium was joined by a private effort at Celera Genomics that used the shotgun sequencing approach and a race to finish the project ensued. With the use of computers the vast number of fragments from Celera were assembled using the framework established by the HGP and in June of 2000, the completion of the draft sequence was announced by the leaders of the public and private projects.
Assembled according to code
DNA is structured as a tightly-associated pair of molecules.[11][21] entwined in the shape of a double helix. The nucleotide repeats contain both the backbone of the molecule, which holds the chain together, and a base, which complements the other DNA strand. The double helix is held together by weak hydrogen bonds between the complementary bases attached to the two strands. If many nucleotides are linked together, as in DNA, the polymer is referred to as a polynucleotide.[22]The nucleotides are very small (just 3.3Å long), but DNA can contain millions of them: the DNA in the largest human chromosome (Chromosome 1) has 220 million base pairs.[23]The DNA chain is 22-26 Å wide (2.2-2.6nm)[24]
The backbone of the DNA strand has alternating phosphate and sugar residues[25]: the sugar is the pentose (five carbon) sugar 2-deoxyribose. The sugar molecules are joined together by phosphate groups that form phosphodiester bonds between the third and fifth carbon atoms in the sugar rings. Because these bonds are asymmetric, a strand of DNA has a 'direction'. In a double helix, the direction of the nucleotides in one strand is opposite to that in the other strand. This arrangement of DNA strands is called antiparallel. The asymmetric ends of a strand of DNA bases are referred to as the 5' (five prime) and 3' (three prime) ends. One of the major differences between DNA and RNA is the sugar: 2-deoxyribose is replaced by ribose in RNA.[21]
The four bases in DNA are adenine (A), cytosine (C), guanine (G) and thymine (T), and these bases are attached to the sugar/phosphate to form the complete nucleotide. Adenine and guanine are fused five- and six-membered heterocyclic compounds called purines, while cytosine and thymine are six-membered rings called pyrimidines.[22] A fifth pyrimidine base, uracil (U), replaces thymine in RNA. Uracil is normally only found in DNA as a breakdown product of cytosine, but bacterial viruses contain uracil in their DNA.[26] In contrast, following synthesis of certain RNA molecules, many uracils are converted to thymines. This occurs mostly on structural and enzymatic RNAs like tRNAs and ribosomal RNA.[27]
The double helix is a right-handed spiral. As the DNA strands wind around each other, gaps (a major and a minor groove) between the two phosphate backbones reveal the sides of the bases inside (see animation). Two of these grooves twist around the surface of the double helix: the major groove is 22 Å wide and the minor groove is 12 Å wide.[28] The edges of the bases are more accessible in the major groove, so proteins like transcription factors that can bind to specific sequences in double-stranded DNA usually make contacts to the sides of the bases exposed in the major groove.[29] For many DNA: protein complexes, up to one-half of the contacts involve the DNA phosphate groups, and thus many DNA binding proteins have more positive amino acids (lysine and arginine) than usual.
The structure of the DNA as a chromosome is formed by a scaffolding of proteins called histones
Base pairing
Each type of base on one strand of DNA forming a bond with a complementary base on the other strand, is called base pairing'. Purines form hydrogen bonds to pyrimidines; for example, A bonds only to T, and C bonds only to G. This arrangement of two nucleotides joined together across the double helix is called a base pair. In a double helix, the two strands are also held together by hydrophobic effects and by a variation of pi stacking.[30] Hydrogen bonds are weak, and can be broken and rejoined quite easily, so the two strands of DNA in a double helix can be pulled apart like a zipper, either by mechanical force or by high temperatures.[31] Because of this complementarity, the information in the double-stranded sequence of a DNA helix is duplicated on each strand. The reversible and specific interaction between complementary base pairs is vital in DNA replication. and is critical for all the functions of DNA in living organisms.[1]
The two types of base pairs form different numbers of hydrogen bonds; AT forms two hydrogen bonds, and GC forms three, so the GC base-pair is stronger than the AT pair. Thus, long DNA helices with a high GC content have strongly interacting strands, while short helices with high AT content have weakly interacting strands.[32] Parts of the DNA double helix that need to separate easily tend to have a high AT content, making the strands easier to pull apart.[33] The strength of this interaction can be measured by finding the temperature required to break the hydrogen bonds (their 'melting temperature'). When all the base pairs in a DNA double helix melt, the strands separate, leaving two single-stranded molecules in solution. Because base-pairing causes changes in the UV-visible spectrum of the bases, DNA melting can be monitored by observing the hypochronicity (or loss thereof) as a function of temperature. These molecules have no single shape, but some conformations are more stable than others.
Flow of information
There are many interactions that happen between DNA and other molecules to coordinate its functions. When cells divide, the genetic information must be duplicated to produce two complementary copies of DNA in a process called DNA replication. When a cell uses the information in a gene, the DNA sequence is copied into a complementary single strand of RNA in a process called transcription. Of the transcribed sequences, some are used to directly make a matching protein sequence by a process called translation (meaning translation from a nucleic acid polymer to an amino acid polymer). The other transcribed RNA sequences may have regulatory, structural or catalytic roles.
Replication
- Further information: Replication of a circular bacterial chromosome
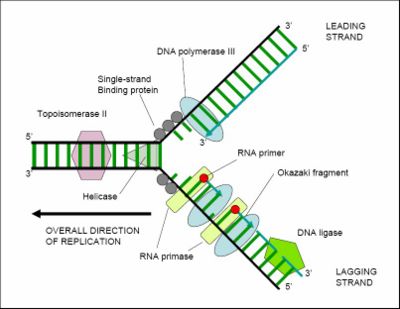
For an organism to grow, its cells must multiply, and this occurs by cell division: one cell splits to create two new 'daughter' cells. When a cell divides it must make an exact copy of its DNA so that the two daughter cells have the same genetic information as the parent cell. DNA replication requires a complex set of proteins, each dedicated to one of several different tasks needed to replicate this large molecule in an orderly and precise fashion. The double-stranded complementary structure of DNA provides a simple mechanism for this replication. The two strands of DNA are separated by a helicase, and then each strand's complementary DNA sequence is recreated by an enzyme called DNA polymerase. This makes the complementary strand by finding the correct base (through complementary base pairing), and bonding it to the original strand. DNA polymerases can only extend a DNA strand in a 5' to 3' direction, so other mechanisms are needed to copy the asymetrical antiparallel strand of the double helix.[34] In this way, the base on the old strand dictates which base appears on the new strand, and the cell ends up, after proofreading, with a perfect copy of its DNA.
Further capacity for DNA replication with substantial rearrangement is provided by mechanisms for DNA movement, inversion, and duplication, illustrated by various mobile DNAs such as transposons and proviruses (which has major implications for understanding mechanisms of molecular evolution).
Helicases
Helicases are a type of 'molecular motor'; they use the chemical energy in nucleoside triphosphates (predominantly ATP) to break hydrogen bonds between bases and unwind the DNA double helix into single strands.[35] Helicases are essential for most processes where enzymes need to access the DNA bases.
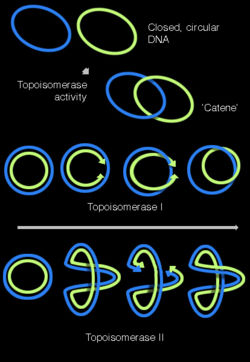
Topoisomerases
When a section of the double helix is being unwoud, the remaining sections are tangled. Topoisomerases have both nuclease and ligase activity, and they can change the amount of supercoiling in DNA. Some work by cutting the DNA helix and allowing one section to rotate; the enzyme then seals the DNA break.[36] Others can cut one DNA helix and then pass a second strand of DNA through this break, before rejoining the helix.[37] Topoisomerases are required for many processes involving DNA, such as DNA replication and transcription.[38]
Polymerases
DNA polymerases copy DNA sequences. Accuracy is very important for this, so many polymerases have a proofreading activity: they can recognizes the occasional mistakes that can occur during synthesis. These enzymes detect the lack of base pairing between mismatched nucleotides, and if a mismatch is detected, a 3′ to 5′ exonuclease activity is activated and the incorrect base is removed.[39] In most organisms, DNA polymerases function in a large complex called the replisome.
Polymerases synthesise polynucleotides from nucleoside triphosphates. They add nucleotides onto the 3′ hydroxyl group of the previous nucleotide in the DNA strand, so all polymerases work in a 5′ to 3′ direction.[40] In the active site of these enzymes, the nucleoside triphosphate substrate base-pairs to a single-stranded polynucleotide template: this allows polymerases to synthesise the complementary strand of this template accurately.
RNA-dependent DNA polymerases copy the sequence of an RNA strand into DNA. One example is reverse transcriptase, which is a viral enzyme involved in the infection of cells by retroviruses; another example is telomerase, which is required for the replication of telomeres.[41][42] Telomerase is an unusual polymerase because it contains its own RNA template as part of its structure.[43]
Transcription
The genetic code consists of three-letter 'words' called codons formed from a sequence of three nucleotides. As there are four bases in three-letter combinations, there are 64 possible codons, and these encode the twenty standard amino acids. For example, the sequences ACU, CAG and UUU in mRNA are translated to threonine, glutamine and phenylalanine, respectively. Many amino acids, have more than one possible codon (for example, ACU and ACA both code for threonine).
Transcription is carried out by a RNA polymerase that copies a DNA sequence into RNA. The enzyme binds to a promoter and separates the DNA strands. It then copies the gene sequence into a mRNA transcript until it reaches a region of DNA called the terminator, where it halts and detaches from the DNA. This complementary copy is then decoded by a ribosome that reads the RNA sequence by base-pairing the mRNA to a specific aminoacyl-tRNA; an amino acid carried by a transfer RNA (tRNA). There is one start codon (AUG, which also encodes for methionine) and three 'stop' or 'nonsense' codons (UAA, UGA and UAG) that signify the end of the coding region.
RNA polymerase II, which transcribes most of the genes in the human genome, operates as part of a large protein complex with multiple regulatory and accessory subunits.[44]
Translation
After a gene that encodes a protein is transcribed to a messenger RNA (mRNA), the code is delivered to an RNA/protein complex, an enzyme, called a ribosome. The ribosome translates the information in the mRNA and assembles the corresponding string of amino acids one by one ending as it moves along the mRNA, ending up with the appropriate polypeptide. Several ribosomes may move along the mRNA simultaneously.
Nature of genes
DNA contains the genetic information that is the basis for living functions including growth, reproduction and evolution. This information is held in segments of the DNA called genes that may span in size from dozens of DNA base-pairs to many thousands of base-pairs. DNA usually occurs as several large, linear chromosomes, each of which may contain hundreds or thousands of genes in eukaryotes. The complete set of chromosomes in a cell makes up its genome; the human genome has about three billion base pairs of DNA arranged into 46 chromosomes [45] and contains 25-30,000 genes.[46] Prokaryotes generally have a single large circular chromosome, but they often possess other miniature chromosomes called plasmids.
All humans have essentially the same DNA, but have slightly different genes; on average, the DNA of two individuals differs at about three million bases. These differences are rarely in the protein-coding sequences of genes, but can affect how particular genes are regulated &mdash, exactly where in the body a gene is expressed, how intensely it is expressed, or how expression is regulated by other genes or by environmental factors. These slight differences help to make every human being unique. By comparison, the genome of our closest living relative, the chimpanzee, differs from the human genome by about 30 million bases. [47]
The elusive definition
Our understanding of the role genes play in cells, starting from abstract concepts of inheritable "factors" whose composition was unknown, has been continually revised throughout the history of genetics. This has led to several definitions of a gene. One way to define a gene is simply as a segment of DNA that is transcribed into RNA - that is - the gene is a unit of transcription. This definition encompasses genes for non-coding RNAs, such as ribosomal RNA (rRNA) and transfer RNA (tRNA), as well as messenger RNA (mRNA) which is used for encoding the sequences of proteins.
A second approach is to define a gene as a region of DNA that encodes a single polypeptide. By this definition, any particular mRNA transcription unit can contain more than one gene, and thus a mRNA can carry regions encoding one or more polypeptides. Such a multi-genic transcription unit is called an operon and are common in prokaryotic cells.
Both these definitions are similar to a "bead on a string" analogy with distinct genes as sequentially ordered units along a chromosome. A more fuzzy definition considers genes as units of biological function. This definition can include cis-acting sequences of DNA that are not transcribed and flank a coding region. They act as specific binding sites for a variety of transcription factor proteins involved with regulating gene expression; examples of such sequences are enhancers, promoters and operators. In this model the "bead on a string" analogy breaks down since the cis-acting factors from different genes do overlap and this is closer to the reality of gene structure in a chromosome.
Expression of genes
How genes are regulated — how they are turned on or off — is an important topic in modern biology, and research continually yields surprising insights into how phenotypic traits and biological adaptations are determined.
In the 1950's, investigations of the bacterium Escherichia coli led to the recognition that the region upstream of a transcribed region provides a place for the enzyme RNA polymerase to attach to DNA and start transcribing RNA in the 5' to 3' direction of the nucleic acid chain. The site at which this occurs came to be called the promoter. Other regulatory proteins (such as repressors) influence transcription by binding to a region near to or overlapping the promoter, called the operator. In the early years of modern genetics, emphasis was given to transcriptional regulation as an important and common means of modulating gene expression, but today we realize that there are a wide range of mechanisms by which the expression of mRNA and proteins can be modulated by both external and internal signals in cells.
Genomes
The human genome has about three billion base pairs of DNA arranged into 46 chromosomes[45], and contains 25-30,000 genes [46], the simple nematode C elegans has almost as many genes (more than 19,000)[48]. In eukaryotes, DNA is located mainly in the cell nucleus (there are also small amounts in mitochondria and chloroplasts). In prokaryotes, the DNA is in an irregularly shaped body in the cytoplasm called the nucleoid.[49] The DNA is usually in linear chromosomes in eukaryotes, and circular chromosomes in prokaryotes. In many species, only a small fraction of the genome encodes protein: only about 1.5% of the human genome consists of protein-coding exons, while over 50% consists of non-coding repetitive sequences.[50] While most of the non-coding elements in human DNA were once considered "junk DNA" with no known function, recent studies suggest a variety of roles for these regions, such as RNA silencing[51] or gene swapping by transposable elements.[52]
Some other vertebrates, including the puffer fish Fugu have more compact genomes, and (for multicellular organisms) there seems to be no consistent relationship between the size of the genome and the complexity of the organism [53]. Some non-coding DNA sequences are now known to have a structural role in chromosomes. In particular, telomeres and centromeres contain few genes, but are important for the function and stability of chromosomes.[54][55] An abundant form of non-coding DNA in humans are pseudogenes, which are copies of genes that have been disabled by mutation;[56] these are usually just molecular 'fossils', but they can provide the raw genetic material for new genes.[57]
A recent challenge to the long-standing view that the human genome consists of relatively few genes along with a vast amount of "junk DNA" comes from the ENCyclopedia Of DNA Elements (ENCODE) consortium.[58][59] Their survey of the human genome shows that most of the DNA is transcribed into molecules of RNA. This broad pattern of transcription was unexpected, but whether these transcribed (but not translated) elements have any biological function is not yet clear.[59]
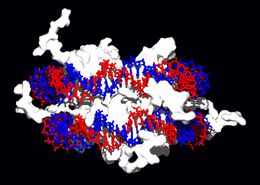
DNA-binding proteins
All of the functions of DNA depend on its interactions with proteins. Some of these interactions are non-specific, others are specific in that the protein can only bind to a particular DNA sequence. Some enzymes can also bind to DNA and of these, the polymerases that copy the DNA base sequence in transcription and DNA replication are particularly important.
|
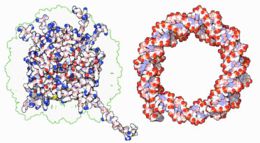
|
Within chromosomes, DNA is held in complexes between DNA and structural proteins, forming a compact structure called chromatin. In eukaryotes, this structure involves DNA binding to small basic proteins called histones, while in prokaryotes many types of proteins are involved.[60] The histones form a disk-shaped complex called a nucleosome which has two complete turns of double-stranded DNA wrapped around it. These interactions are formed through basic residues in the histones making ionic bonds to the acidic sugar-phosphate backbone of the DNA, and are largely independent of the base sequence.[61] Chemical modifications of these basic amino acid residues include methylation, phosphorylation and acetylation.[62] These changes alter the strength of the interaction between the DNA and the histones, making the DNA more or less accessible to transcription factors and changing the rate of transcription.[63] Other non-specific DNA-binding proteins found in chromatin include the high-mobility group proteins, which bind preferentially to bent or distorted DNA.[64] These proteins are important in bending arrays of nucleosomes and arranging them into more complex chromatin structures.[65]
Another group of DNA-binding proteins bind single-stranded DNA. In humans, replication protein A is the best-characterised member of this family, and is essential for most processes where the double helix is separated (including DNA replication, recombination and DNA repair).[66] These proteins seem to stabilize single-stranded DNA and protect it from forming stem loops or being degraded by nucleases.
Other proteins bind to particular DNA sequences. The most intensively studied of these are the transcription factors. Each of these proteins binds to a particular set of DNA sequences and thereby activates or inhibits the transcription of genes with these sequences close to their promoters. Transcription factors do this in two ways. Some can bind the RNA polymerase responsible for transcription, either directly or through other mediator proteins; this locates the polymerase at the promoter and allows it to begin transcription.[67] Other transcription factors can bind enzymes that modify the histones at the promoter, this will change the accessibility of the DNA template to the polymerase.[68]
These DNA targets can occur throughout an organism's genome, so changes in the activity of one type of transcription factor in a given cell can affect the expression of many genes in that cell.[69] Consequently, these proteins are often the targets of the signal transduction processes that mediate responses to environmental changes or cellular differentiation and development. The specificity of transcription factor interactions with DNA arises because the proteins make multiple contacts to the edges of the DNA bases, allowing them to "read" the DNA sequence. Most of these interactions occur in the major groove, where the bases are most accessible.[29]
DNA methylation
The expression of genes is influenced by the chromatin structure of a chromosome, and regions of heterochromatin (with little or no gene expression) correlate with the methylation of cytosine.[70] These structural changes to the DNA are one type of epigenetic change that can alter chromatin structure, and they are inheritable. Epigenetics refers to features of organisms that are stable over successive rounds of cell division but which do not involve changes in the underlying DNA sequence[71]. Epigenetic changes are important in cellular differentiation, allowing cells to maintain different characteristics despite containing the same genomic material. Some epigenetic features can be inherited from one generation to the next.[72]
The level of methylation varies between organisms; the nematode C. elegans lacks any cytosine methylation, while up to 1% of the DNA of vertebrates contains 5-methylcytosine.[73] Despite the biological importance of 5-methylcytosine, it is susceptible to spontaneous deamination, and methylated cytosines are therefore mutation 'hotspots'.[74] Other base modifications include adenine methylation in bacteria and the glycosylation of uracil to produce the "J-base" in kinetoplastids.[75][76]
The good, the neutral and the ugly
Genetic recombination
A DNA helix does not usually interact with other segments of DNA, and in human cells the different chromosomes even occupy different regions of the nucleus (called "chromosome territories").[77] This physical separation of chromosomes is important for the ability of DNA to function as a stable repository for information, as one of the few times chromosomes interact is when they recombine. Recombination is when two DNA helices break, swap a section and then rejoin. In eukaryotes, this usually occurs during meiosis, when two chromatids are paired together in the center of the cell. This allows chromosomes to exchange genetic information and produces new combinations of genes.[78] Genetic recombination can also be involved in DNA repair.[79] The most common form of recombination is homologous recombination, where the two chromosomes involved share very similar sequences. However, recombination can also damage cells, by producing chromosomal translocations and genetic abnormalities. Recombination reactions are catalyzed by recombinases,[80] which have a DNA-dependent ATPase activity. The recombinase makes a 'nick' in one strand of a DNA double helix, allowing the nicked strand to separate from its complementary strand and anneal to one strand of the double helix on the opposite chromatid. A second nick allows the strand in the second chromatid to separate and anneal to the strand in the first helix, forming a cross-strand exchange (also called a Holliday junction). The Holliday junction is a tetrahedral junction structure which can be moved along the pair of chromosomes, swapping one strand for another. The recombination is then halted by cleavage of the junction and re-ligation of the released DNA.[81]
Nucleases and ligases
Nucleases cut DNA strands by catalyzing the hydrolysis of the phosphodiester bonds (nucleases that hydrolyse nucleotides from the ends of DNA strands are called exonucleases, while endonucleases cut within strands). The most frequently-used nucleases in molecular biology are the restriction endonucleases, which cut DNA at specific sequences. For instance, the EcoRV enzyme recognizes the 6-base sequence 5′-GAT|ATC-3′ and makes a cut at the vertical line. In nature, these enzymes protect bacteria against phage infection by digesting the phage DNA when it enters the bacterial cell, acting as part of the restriction modification system.[82] In technology, these sequence-specific nucleases are used in molecular cloning and DNA fingerprinting.
DNA ligases can rejoin cut or broken DNA strands, using the energy from either adenosine triphosphate or nicotinamide adenine dinucleotide. Ligases are particularly important in lagging strand DNA replication, as they join together the short segments of DNA produced at the replication fork into a complete copy of the DNA template. They are also used in DNA repair and genetic recombination.[83]
Mutations
DNA can be damaged by many different agents. Mutagens are agents which can produce genetic mutations - these are alterations of one DNA base to another base. Examples of mutagens include oxidizing agents, alkylating agents and high-energy electromagnetic radiation such as ultraviolet light and x-rays. Lesions of DNA, in which residues are changed to a structure that is not a normal feature of DNA, are different from mutations. However, damaged DNA can give rise to mutations, and indeed, some DNA repair processes are error prone and thus themselves generate mutations. Many mutations have no effect on the function of the genes but some may cause a change in gene expression and function leading to a new phenotype.
Ultraviolet radiation damages DNA mostly by producing thymine dimers.[84] Oxidants such as free radicals or hydrogen peroxide can cause several forms of damage, including base modifications (particularly of guanosine) as well as double-strand breaks.[85] It has been estimated that, in each human cell, about 500 bases suffer oxidative damage every day.[86][87] Of these lesions, the most damaging are double-strand breaks, as they can produce point mutations, insertions and deletions from the DNA sequence, as well as chromosomal translocations.[88]
Many mutagens intercalate into the space between two adjacent base pairs. These are mostly polycyclic, aromatic, and planar molecules, and include ethidium, proflavin, daunomycin, doxorubicin and thalidomide. For an intercalator to fit between base pairs, the bases must separate, distorting the DNA strand by unwinding of the double helix. These structural modifications inhibit transcription and replication processes, causing both toxicity and mutations. As a result, DNA intercalators are often carcinogens. Nevertheless, because they can inhibit DNA transcription and replication, they are also used in chemotherapy to inhibit DNA-replication in rapidly-growing cancer cells.[89]
DNA Repair
Environmental factors such as heat and ultraviolet radiation can produce hundreds and even thousands of mistakes in one day. It is crucial that DNA replication accuracy is maintained. Repair is made possible by the complementary structure of DNA. A diverse set of more than 50 repair mechanisms are available to the cell to correct the mistakes. Replication error, after proofreading, is estimated to be one error in 10,000,000,000 replications. [90]
An insight into evolution
As well as being susceptible to largely random mutations (that usually affect just a single base), some regions of DNA are specialised to undergo dramatic, rapid, non-random rearrangements, or to undergo more subtle changes at a high frequency so that the expression of a gene is dramatically altered. Such rearrangements include various versions of site-specific recombination. This depends on enzymes that recognise particular sites on DNA and create novel structures such as insertions, deletions and inversions. For instance, DNA regions in the small genome of the bacterial virus P1 can invert, enabling different versions of tail fibers to be expressed in different viruses. Similarly, site-specific recombinase enzymes are responsible for mating type variation in yeast, and for flagellum type (phase) changes in the bacterium Salmonella enterica Typhimurium.
More subtle mutations can occur in micro-satellite repeats (also called homopolymeric tracts of DNA). An example of such a structure are short DNA intervals where the same base is tandemly repeated, as in 5'-gcAAAAAAAAAAAttg-3', or a dinucleotide or even a triplet as in 5'- ATGATGATGATGATGATGATG-3'. DNA polymerase III is prone to make 'stuttering errors' at such repeats. As a consequence, changes in repeat number occur quite often during cell replication, and when they appear at a position where the spacing of nucleotide residues is critical for gene function, they can cause changes to the phenotype.
The stomach ulcer bacterium Helicobacter pylori is a good example of how homopolymeric tracts can enable quasi-directed evolution of particular genes. H. pylori has 46 genes that contain homopolymeric runs of nucleotides or dinucleotide repeats that are prone to frequent length changes as a consequence of stuttering errors during replication. These changes can lead to frequent reversible inactivation of these genes, or to changed gene transcription if the repeat is located in a regulatory sequence. This generates highly diverse populations of H. pylori in an individual human host, and this diversity helps the bacterium evade the immune system.[91]
Triplet repeats behave similarly, but are particularly suited to evolution of proteins with differing characteristics. In the clock-like period gene of the fruit fly, triplet repeats 'fine tune' an insect's biological clock in response to changes in environmental temperatures. Triplet repeats are widely distributed in genomes, and their high frequency of mutation is responsible for several genetically determined disorders in humans.[92]
Impact on society
Forensics
Forensic scientists can use DNA in blood, semen, skin, saliva or hair to match samples collected at a crime scene to samples taken from possible suspects. This process is called genetic fingerprinting or more formally, DNA profiling. In DNA profiling, the lengths of variable sections of repetitive DNA (such as short tandem repeats and minisatellites) are compared between people. This is usually very reliable for identifying the source of a sample,[93] but identification can be complicated if the samples that are collected include DNA from several people.[94] DNA profiling was developed in 1984 by British geneticist Sir Alec Jeffreys,[95] and first used in forensic science to convict Colin Pitchfork (and to clear the prime suspect) in the 1988 Enderby murders case.[96] People convicted of certain types of crimes may be required to provide a sample of DNA for a database. This has helped investigators solve old cases where only a DNA sample was obtained from the scene. DNA profiling can also be used to identify victims of mass casualty incidents.[97]
Bioinformatics
One branch of bioinformatics involves the analysis of DNA sequence data; DNA from hundreds of different organisms has now been partially sequenced, and this information is stored in massive databases. The development of techniques to store and search DNA sequences has led to many applications in computer science.[98] String-searching or 'matching' algorithms, which identify a given sequence of letters inside a larger sequence of letters, are used to search for specific sequences of nucleotides. The related problem of sequence alignment aims to identify homologous sequences; these are sequences that are very similar but not identical. When two different genes in an organism have very similar sequences, this is evidence that, at some stage in evolution, a single gene was duplicated, and the sequences subsequently diverged (under different selection pressures) by incorporating different mutations. Identifying such holologies can give valuable clues about the likely function of novel genes. Similarly, identifying homologies between genes in different organisms can be used to reconstruct the evolutionary relationships between organisms.[99] Data sets representing entire genomes' worth of DNA sequences, such as those produced by the Human Genome Project, are difficult to use without annotations, which label the locations of genes and regulatory elements on each chromosome. Regions of DNA sequence that have patterns that are characteristic of protein- or RNA-coding genes can be identified by gene finding algorithms, allowing researchers to predict the presence of particular gene products in an organism.[100]
Molecular cloning
The small chromosomes of bacteria have proved extraordinarily useful for analysing genetic mechanisms and genome structure in many organisms, including humans. This utility arose because of several important discoveries made by microbiologists in the 1950-70's that provided new laboratory tools for directly manipulating genes. The principal laboratory tools and techniques discovered in this period were:
- Compact circular plasmid molecules such as ColE1 that were relatively easy to extract and purify from bacterial cells, and when inserted back inside living bacterial cells could serve as genetically stable carriers (vectors) of novel DNA fragments. These vectors were propagated in bacterial cell lines termed "clones". Since each plasmid vector in these clones carried a single DNA molecular fragment, these could be considered to be 'molecular clones'.
- Convenient methods for extracting plasmid DNA from cells, physical analysis of this DNA, and reinsertion of circular plasmid DNA back inside living cells. The main method for DNA re-insertion is termed DNA transformation - that is, direct uptake of naked DNA molecules by cells. One technique that assisted DNA transformation was the use of selectable genetic markers, and plasmid-borne bacterial antibiotic resistance provided such markers in the form of traits like ampicillin resistance and tetracycline resistance.
- Restriction endonucleases such as the enzymes EcoRI or HindIII, were found to be useful for specifically digesting DNA at particular sites and creating novel combinations of DNA fragments in the laboratory.
- Restriction enzymes enabled re-annealing different DNA fragments, such as circular plasmid DNA which had been linearised at a single EcoRI restriction endonuclease target site together with another fragment, say an EcoRI generated fragment of a human chromosome. By sealing together two such fragments with the enzyme DNA ligase, novel hybrid-plasmid molecules could be created in which "foreign" DNA inserts were carried in a chimeric or hybrid plasmid, often called a recombinant DNA molecule.
After the mid-1970's, re-insertion of recombinant DNA plasmids into living bacterial cells, such as those of Escherichia coli opened up many new approaches in biotechnology. These made it possible to manufacture, for example, human hormones such as insulin in microbial cell-based protein factories. When combined with other genetic techniques, the field of "recombinant DNA technology" opened up the possibility of decoding the gene sequence of whole genomes. This latter area is now usually referred to as genomics, and includes the Human Genome Project.
Bringing it together
On January 28, 2008, an international research consortium announced the 1000 Genomes Project involving sequencing the genomes of at least a thousand people from around the world to create the most detailed and medically useful picture to date of human genetic variation. Drawing on the expertise of multidisciplinary research teams, the 1000 Genomes Project will develop a new map of the human genome that will provide a view of biomedically relevant DNA variations at a resolution unmatched by current resources. “The 1000 Genomes Project will examine the human genome at a level of detail that no one has done before,” said Richard Durbin, Ph.D., of the Wellcome Trust Sanger Institute, who is co-chair of the consortium. “Such a project would have been unthinkable only two years ago. Today, thanks to amazing strides in sequencing technology, bioinformatics and population genomics, it is now within our grasp. So we are moving forward to build a tool that will greatly expand and further accelerate efforts to find more of the genetic factors involved in human health and disease.”
As with other major human genome reference projects, data from the 1000 Genomes Project will be available to the worldwide scientific community through freely accessible public databases.
One example of the impact on understanding the biological basis for schizophrenia, was reported in Scientific American.[101]
The researchers determined which affected genes were likely to cause damage—and where in the body that damage that might occur. The flawed genes in the schizophrenia patients were overwhelmingly linked to changes in pathways responsible for communication between and within nerve cells. In fact, one gene, ERBB4, is known to code for a receptor that interacts with neuregulin 1, a protein that's been associated with the illness for a decade. "The silver lining is that these new findings show us … that we were already on the right track," Sebat says."
References
- ↑ 1.0 1.1 Alberts, Bruce; Alexander Johnson, Julian Lewis, Martin Raff, Keith Roberts, and Peter Walters (2002). Molecular Biology of the Cell; Fourth Edition. New York and London: Garland Science. ISBN 0-8153-3218-1.
- ↑ Kornberg, Arthur. DNA Replication. W.H.Freeman and Co. (1980) p13
- ↑ Dahm R (2005). "Friedrich Miescher and the discovery of DNA". Dev Biol 278: 274-88. PMID 15680349.
- ↑ Levene P (1919). "The structure of yeast nucleic acid". J Biol Chem 40: 415-24.
- ↑ Avery O, MacLeod C, McCarty M (1979). "Studies on the chemical nature of the substance inducing transformation of pneumococcal types. Inductions of transformation by a desoxyribonucleic acid fraction isolated from pneumococcus type III". J Exp Med 149 (2): 297-326. PMID 33226.
- ↑ Asimov, Isaac. (1962) The Genetic Code. pg viii New American Library. Written while he was Associate Professor of Biochemistry at Boston University School of Medicine. "It launched in a new direction the investigation into the nature of life and brought about a new research methods. The branch of science now called "molecular biology" came into its own."
- ↑ Hershey A, Chase M (1952). "Independent functions of viral protein and nucleic acid in growth of bacteriophage". J Gen Physiol 36 (1): 39-56. PMID 12981234.
- ↑ Interview of Chargaff at DNA interactive web site.
- ↑ Astbury W (1947). "Nucleic acid". Symp Soc Exp Biol 1.
- ↑ 10.0 10.1 Watson JD, Crick FHC (1953) A structure for deoxyribose nucleic acid. 'Nature 171:737–8 Accessed 13 Feb 2007.
- ↑ 11.0 11.1 Watson J, Crick F (1953). "Molecular structure of nucleic acids; a structure for deoxyribose nucleic acid". Nature 171: 737-8. PMID 13054692.
- ↑ Nature Archives Double Helix of DNA: 50 Years
- ↑ Franklin R, Gosling RG (1953)Molecular configuration in sodium thymonucleate. Nature N 171, 740–1
- ↑ Original X--ray diffraction image
- ↑ Wilkins MHF, Stokes AR, Wilson HR (1953) Molecular structure of deoxypentose nucleic acids.Nature [http://www.nature.com/nature/dna50/wilkins.pdf 171:738–40
- ↑ Franklin R, Gosling RG (1953) Evidence for 2-chain helix in crystalline structure of Sodium Deoxyribonucleate. Nature 172:156–7
- ↑ The Nobel Prize in Physiology or Medicine 1962 Nobelprize .org Accessed 22 Dec 06
- ↑ Crick FHC (1955) On degenerate templates and the adaptor hypothesis(PDF) genome.wellcome.ac.uk (Accessed 22 Dec 2006
- ↑ Meselson M, Stahl F (1958). "The replication of DNA in Escherichia coli". Proc Natl Acad Sci USA 44: 671-82. PMID 16590258.
- ↑ The Nobel Prize in Physiology or Medicine 1968 Nobelprize.org Accessed 22 Dec 06
- ↑ 21.0 21.1 Berg J et al. (2002) Biochemistry. WH Freeman and Co. ISBN 0-7167-4955-6
- ↑ 22.0 22.1 Abbreviations and Symbols for Nucleic Acids, Polynucleotides and their Constituents IUPAC-IUB Commission on Biochemical Nomenclature (CBN) Accessed 03 Jan 2006
- ↑ Gregory S et al. (2006). "The DNA sequence and biological annotation of human chromosome 1". Nature 441: 315-21. PMID 16710414.
- ↑ Mandelkern M et al. (1981). "The dimensions of DNA in solution". J Mol Biol 152: 153-61. PMID 7338906.
- ↑ Ghosh A, Bansal M (2003). "A glossary of DNA structures from A to Z". Acta Crystallogr D Biol Crystallogr 59: 620-6. PMID 12657780.
- ↑ Takahashi I, Marmur J (1963). "Replacement of thymidylic acid by deoxyuridylic acid in the deoxyribonucleic acid of a transducing phage for Bacillus subtilis". Nature 197: 794-5. PMID 13980287.
- ↑ Agris P (2004). "Decoding the genome: a modified view". Nucleic Acids Res 32: 223 – 38. PMID 14715921.
- ↑ Wing R et al. (1980). "Crystal structure analysis of a complete turn of B-DNA". Nature 287: 755–8. PMID 7432492.
- ↑ 29.0 29.1 Pabo C, Sauer R. "Protein-DNA recognition". Ann Rev Biochem 53: 293-321. PMID 6236744.
- ↑ Ponnuswamy P, Gromiha M (1994). "On the conformational stability of oligonucleotide duplexes and tRNA molecules". J Theor Biol 169: 419-32. PMID 7526075.
- ↑ Clausen-Schaumann H et al. (2000). "Mechanical stability of single DNA molecules". Biophys J 78: 1997-2007. PMID 10733978.
- ↑ Chalikian T et al. (1999). "A more unified picture for the thermodynamics of nucleic acid duplex melting: a characterization by calorimetric and volumetric techniques". Proc Natl Acad Sci USA 96: 7853-8. PMID 10393911.
- ↑ deHaseth P, Helmann J (1995). "Open complex formation by Escherichia coli RNA polymerase: the mechanism of polymerase-induced strand separation of double helical DNA". Mol Microbiol 16: 817-24. PMID 7476180.
- ↑ Albà M (2001). "Replicative DNA polymerases". Genome Biol 2: REVIEWS3002. PMID 11178285.
- ↑ Tuteja N, Tuteja R (2004). "Unraveling DNA helicases. Motif, structure, mechanism and function". Eur J Biochem 271: 1849–63. PMID 15128295.
- ↑ Champoux J. "DNA topoisomerases: structure, function, and mechanism". Ann Rev Biochem 70: 369–413. PMID 11395412.
- ↑ Schoeffler A, Berger J (2005). "Recent advances in understanding structure-function relationships in the type II topoisomerase mechanism". Biochem Soc Trans 33: 1465–70. PMID 16246147.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedWang - ↑ Hubscher U et al.. "Eukaryotic DNA polymerases". Annu Rev Biochem 71: 133–63. PMID 12045093.
- ↑ Joyce C, Steitz T (1995). "Polymerase structures and function: variations on a theme?". J Bacteriol 177: 6321–9. PMID 7592405.
- ↑ Tarrago-Litvak L et al. (1994). "The reverse transcriptase of HIV-1: from enzymology to therapeutic intervention". FASEB J 8: 497–503. PMID 7514143.
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedGreider - ↑ Nugent C, Lundblad V (1998). "The telomerase reverse transcriptase: components and regulation". Genes Dev 12 (8): 1073–85. PMID 9553037.
- ↑ Martinez E (2002). "Multi-protein complexes in eukaryotic gene transcription". Plant Mol Biol 50: 925–47. PMID 12516863.
- ↑ 45.0 45.1 Venter J et al. (2001). "The sequence of the human genome". Science 291: 1304–51. PMID 11181995.
- ↑ 46.0 46.1 Human Genome Project Information [1]
- ↑ Pollard KS et al. (2006) Forces shaping the fastest evolving regions in the human genome. PLoS Genet 2:e168 PMID 17040131
- ↑ The C. elegans Sequencing Consortium (1998)Genome Sequence of the Nematode C. elegans: A Platform for Investigating Biology. Science 282: 2012-8 [2]
- ↑ Thanbichler M et al. (2005). "The bacterial nucleoid: a highly organized and dynamic structure". J Cell Biochem 96: 506–21. PMID 15988757.
- ↑ Wolfsberg T et al. (2001). "Guide to the draft human genome". Nature 409: 824-6. PMID 11236998.
- ↑ Volpe, TA "et al." (2002). "Regulation of heterochromatic silencing and histone H3 lysine-9 methylation by RNAi". Science 297: 1833-1837.
- ↑ Biemont, C and Vieira, C (2006). "Genetics: Junk DNA as an evolutionary force". Nature 443: 521-524.
- ↑ See Sydney Brennar's Nobel lecture (2002) [3]
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedwright - ↑ Pidoux A, Allshire R (2005). "The role of heterochromatin in centromere function". Philos Trans R Soc Lond B 360: 569-79. PMID 15905142.
- ↑ Harrison P et al. (2002). "Molecular fossils in the human genome: identification and analysis of the pseudogenes in chromosomes 21 and 22". Genome Res 12: 272-80. PMID 11827946.
- ↑ Harrison P, Gerstein M (2002). "Studying genomes through the aeons: protein families, pseudogenes and proteome evolution". J Mol Biol 318: 1155-74. PMID 12083509.
- ↑ Weinstock GM (2007). "ENCODE: More genomic empowerment". Genome Research 17: 667-668. PMID 17567987.
- ↑ 59.0 59.1 ENCODE Project Consortium (2007). "Identification and analysis of functional elements in 1% of the human genome by the ENCODE pilot project". Nature 447: 799-816. PMID 17571346.
- ↑ Sandman K et al. (1998). "Diversity of prokaryotic chromosomal proteins and the origin of the nucleosome". Cell Mol Life Sci 54: 1350-64. PMID 9893710.
- ↑ Luger K et al. (1997). "Crystal structure of the nucleosome core particle at 2.8 A resolution". Nature 389: 251-60. PMID 9305837.
- ↑ Jenuwein T, Allis C (2001). "Translating the histone code". Science 293: 1074-80. PMID 11498575.
- ↑ Ito T. "Nucleosome assembly and remodelling". Curr Top Microbiol Immunol 274: 1-22. PMID 12596902.
- ↑ Thomas J (2001). "HMG1 and 2: architectural DNA-binding proteins". Biochem Soc Trans 29: 395-401. PMID 11497996.
- ↑ Grosschedl R et al. (1994). "HMG domain proteins: architectural elements in the assembly of nucleoprotein structures". Trends Genet 10: 94-100. PMID 8178371.
- ↑ Iftode C et al. (1999). "Replication protein A (RPA): the eukaryotic SSB". Crit Rev Biochem Mol Biol 34: 141-80. PMID 10473346.
- ↑ Myers L, Kornberg R. "Mediator of transcriptional regulation". Ann Rev Biochem 69: 729-49. PMID 10966474.
- ↑ Spiegelman B, Heinrich R (2004). "Biological control through regulated transcriptional coactivators". Cell 119: 157-67. PMID 15479634.
- ↑ Li Z et al. (2003). "A global transcriptional regulatory role for c-Myc in Burkitt's lymphoma cells". Proc Natl Acad Sci USA 100: 8164-9. PMID 12808131.
- ↑ For example, cytosine methylation, to produce 5-methylcytosine, is important for X-chromosome inactivation. Klose R, Bird A (2006). "Genomic DNA methylation: the mark and its mediators". Trends Biochem Sci 31: 89–97. PMID 16403636.
- ↑ Bird A (2007). "Perceptions of epigenetics". Nature 447: 396-8. PMID 17522671
- ↑ Chandler VL (2007). "Paramutation: from maize to mice". Cell 128: 641-5.
- ↑ Bird A (2002). "DNA methylation patterns and epigenetic memory". Genes Dev 16: 6–21. PMID 11782440.
- ↑ Walsh C, Xu G. "Cytosine methylation and DNA repair". Curr Top Microbiol Immunol 301: 283–315. PMID 16570853.
- ↑ Ratel D et al. (2006). "N6-methyladenine: the other methylated base of DNA". Bioessays 28: 309–15. PMID 16479578.
- ↑ Gommers-Ampt J et al. (1993). "beta-D-glucosyl-hydroxymethyluracil: a novel modified base present in the DNA of the parasitic protozoan T. brucei". Cell 75: 1129–36. PMID 8261512.
- ↑ Cremer T, Cremer C (2001). "Chromosome territories, nuclear architecture and gene regulation in mammalian cells". Nat Rev Genet 2: 292-301. PMID 11283701.
- ↑ Pál C et al. (2006). "An integrated view of protein evolution". Nat Rev Genet 7: 337-48. PMID 16619049.
- ↑ O'Driscoll M, Jeggo P (2006). "The role of double-strand break repair - insights from human genetics". Nat Rev Genet 7: 45-54. PMID 16369571.
- ↑ Sung P et al. (2003). "Rad51 recombinase and recombination mediators". J Biol Chem 278: 42729-32. PMID 12912992.
- ↑ Dickman M et al. (2002). "The RuvABC resolvasome". Eur J Biochem 269: 5492-501. PMID 12423347.
- ↑ Bickle T, Krüger D (1993). "Biology of DNA restriction". Microbiol Rev 57: 434–50. PMID 8336674.
- ↑ Doherty A, Suh S (2000). "Structural and mechanistic conservation in DNA ligases.". Nucleic Acids Res 28: 4051–8. PMID 11058099.
- ↑ Douki T et al. (2003). "Bipyrimidine photoproducts rather than oxidative lesions are the main type of DNA damage involved in the genotoxic effect of solar ultraviolet radiation". Biochemistry 42: 9221-6. PMID 12885257.
- ↑ Cadet J et al. (1999). "Hydroxyl radicals and DNA base damage". Mutat Res 424 (1-2): 9-21. PMID 10064846.
- ↑ Shigenaga M et al. (1989). "Urinary 8-hydroxy-2'-deoxyguanosine as a biological marker of in vivo oxidative DNA damage". Proc Natl Acad Sci USA 86: 9697-701. PMID 2602371.
- ↑ Cathcart R et al. (1984). "Thymine glycol and thymidine glycol in human and rat urine: a possible assay for oxidative DNA damage". Proc Natl Acad Sci USA 81: 5633-7. PMID 6592579.
- ↑ Valerie K, Povirk L (2003). "Regulation and mechanisms of mammalian double-strand break repair". Oncogene 22: 5792-812. PMID 12947387.
- ↑ Braña M et al. (2001). "Intercalators as anticancer drugs". Curr Pharm Des 7: 1745-80. PMID 11562309.
- ↑ Hanawalt, P.C., and Setlow, R.B. (eds) Molecular Mechanisms for Repair of DNA. Plenum Press New York.
- ↑ Suerbaum S, Josenhans C (2007) Helicobacter pylori evolution and phenotypic diversification in a changing host. Nat Rev Microbiol 5:441-52 PMID 17505524
- ↑ Christopher Wills discusses the evolutionary significance of these concepts in The Runaway Brain: The Evolution of Human Uniqueness ISBN 0-00-654672-2 (1995)
- ↑ Collins A, Morton N (1994). "Likelihood ratios for DNA identification". Proc Natl Acad Sci USA 91: 6007-11. PMID 8016106.
- ↑ Weir B et al. (1997). "Interpreting DNA mixtures". J Forensic Sci 42: 213-22. PMID 9068179.
- ↑ Jeffreys A et al. (1985). "Individual-specific 'fingerprints' of human DNA.". Nature 316: 76-9. PMID 2989708.
- ↑ Colin Pitchfork - first murder conviction on DNA evidence also clears the prime suspect Forensic Science Service Accessed 23 Dec 2006
- ↑ DNA Identification in Mass Fatality Incidents. National Institute of Justice (September 2006).
- ↑ Baldi, Pierre. Brunak, Soren. Bioinformatics: The Machine Learning Approach MIT Press (2001) ISBN 978-0-262-02506-5
- ↑ Sjölander K (2004). "Phylogenomic inference of protein molecular function: advances and challenges". Bioinformatics 20: 170-9. PMID 14734307.
- ↑ Mount DM (2004). Bioinformatics: Sequence and Genome Analysis, 2. Cold Spring Harbor Laboratory Press. ISBN 0879697121.
- ↑ Nikhil Swaminathan, A New, Genetic Model for Schizophrenia Scientific American 2008, March 28.